Context:
In a groundbreaking development, India's first CAR T-cell therapy has shown remarkable efficacy in treating specific types of blood cancer. The clinical trial results, published in the esteemed medical journal The Lancet, reveal that the therapy was successful in nearly 73% of patients.
What is CAR T-Cell Therapy?
CAR T-cell therapy, also known as chimeric antigen receptor T-cell therapy, is a pioneering approach that harnesses the power of the immune system to combat cancer.
· This treatment involves training the body’s immune cells, known as T-cells, to identify and destroy cancer cells.
· Specifically designed for certain types of blood cancer, CAR T-cell therapy is administered to patients whose cancer has either relapsed or not responded to initial treatment.
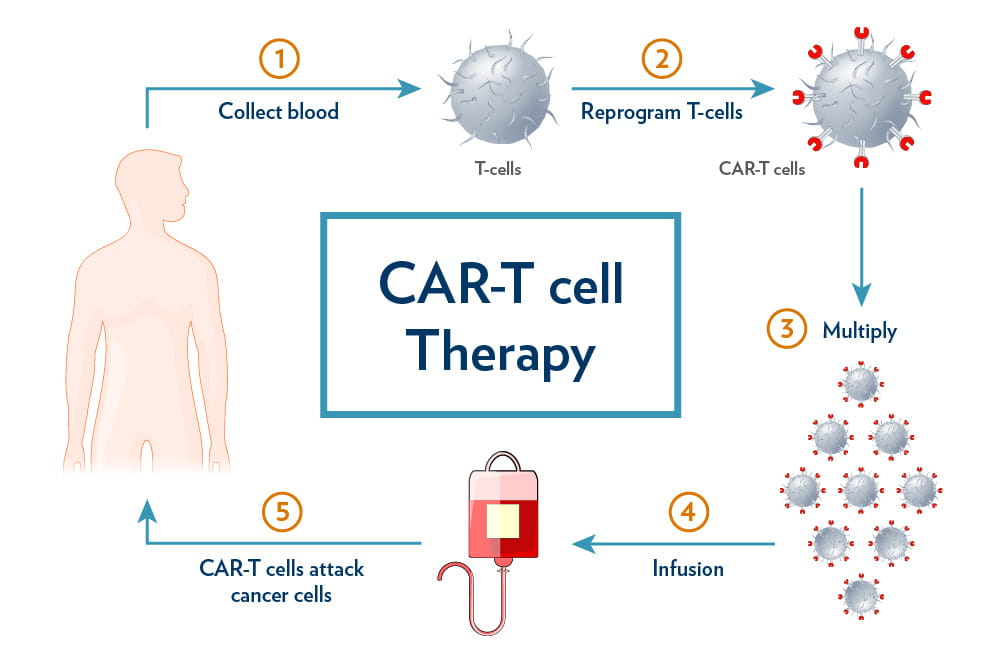
How Does the Therapy Work?
The process begins with the collection of a patient's immune T-cells through blood filtration. These cells are then genetically engineered in a laboratory to add receptors that can bind with cancer cells.
· The modified cells are subsequently multiplied and infused back into the patient. This innovative approach enables the T-cells to recognize and target cancer cells, which would otherwise evade the immune system.
· This personalized treatment has shown significant promise, particularly in treating cancers like acute lymphoblastic leukaemia (ALL) and large B-cell lymphoma.
India’s CAR T-Cell Therapy Breakthrough
India's first clinical trial using CAR T-cell therapy has focused on patients with two types of blood cancers that affect B cells—acute lymphoblastic leukaemia (ALL) and large B-cell lymphoma.
The results have been promising, with nearly 73% of participants showing a positive response to the treatment. This success highlights not only the potential of CAR T-cell therapy in treating blood cancers but also marks a significant step in advancing cancer care in India.
Side Effects observed:
· Hyperinflammation and Organ Damage: A serious immune overreaction, known as cytokine release syndrome (CRS), was observed in 12% of participants. This severe reaction can lead to organ damage and, in some cases, can be fatal.
· Low Red Blood Cell Count: 61% of participants experienced fatigue and weakness due to a low red blood cell count.
· Thrombocytopenia: 65% of patients reported a low platelet count, increasing the risk of bleeding.
· Neutropenia: 96% of participants experienced a low neutrophil count, raising their vulnerability to infections.
Conclusion:
The success of CAR T-cell therapy in India holds the potential to make CAR T-cell therapy more accessible and affordable, contributing to the global fight against cancer. As research continues to advance, it is essential to address the challenges associated with CAR T-cell therapy and make this innovative treatment available to those in need.







